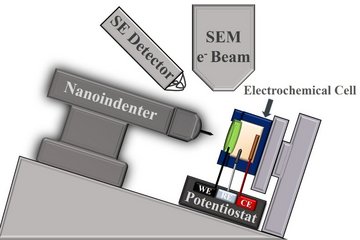
Coupling of SFC to downstream analytics (ICP-MS / UV-Vis)
The investigation of the properties of materials for electrocatalytic applications must be reliable, efficient, fast and inexpensive. The testing of catalysts in real systems or in conventional electrochemical setups, however, requires large amount of catalytic material and/or too much time for electrode preparation. Therefore our group is focused on design and development of high-throughput combinatorial screening tools, which play a major role in the installation of the modern electrochemical methodologies of the group [1]. Moreover, the complementary techniques are incorporated effectively by utilizing sophisticated automation and synchronization of the experimental setup, as well as advanced methods for data evaluation and processing. The automation principle is implemented using a modular software application written in LabVIEW, which allows simultaneous control over different hardware components [2]. With this approach, a large number of samples can be systematically investigated in shorter period of time; while the reliability and reproducibility drastically increases.
The Scanning Flow Cell (SFC) is advanced electrochemical technique based on the concept of channel electrode, where the electrolyte is continuously flowing over the sample. In case of SFC, the working electrode is not integrated in the channels, but externally introduced on three-dimensional allowing quick and efficient exchange of the substrate [1]. Unfortunately, the degradation phenomena cannot be always distinguished and quantified from the electrochemical signal. In order to resolve this challenge, the SFC is coupled with post-analytical techniques like ICP-MS or UV-Vis [3,4]. With respect to the system of interest, different approaches can be used. UV-Vis analysis can provide quantitative and qualitative description of the dissolution rate by using a selective complexing agent and monitoring changes in the adsorption spectra. This methodology was successfully applied for several zinc-based materials [4]. Furthermore, the unique coupling with Inductively Coupled Plasma Mass Spectrometer (ICP-MS), give us a powerful method for online determination of the corrosion products with low degradation rates [5,6,7]. This becomes beneficial in case of the noble metals and their alloys, where the dissolution rates are in the order of a few percent of a single monolayer. Additionally, the synchronization of the methods and sophisticated data evaluation is indispensable for the high-throughput approach in this case. All of this provides a solid basis for the thorough investigations of various material classes reaching from noble metals to steel samples, from a fundamental as well as an applied point of view.
The investigation of the properties of materials for electrocatalytic applications must be reliable, efficient, fast and inexpensive. The testing of catalysts in real systems or in conventional electrochemical setups, however, requires large amount of catalytic material and/or too much time for electrode preparation. Therefore our group is focused on design and development of high-throughput combinatorial screening tools, which play a major role in the installation of the modern electrochemical methodologies of the group [2]. Moreover, the complementary techniques are incorporated effectively by utilizing sophisticated automation and synchronization of the experimental setup, as well as advanced methods for data evaluation and processing.