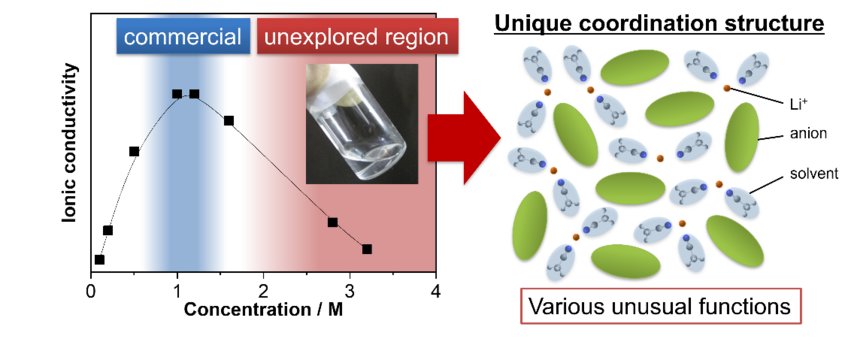
Salt-concentrated liquid electrolytes: unique features and battery applications
An ever-increasing demand for better batteries (with high voltage, high capacity, fast charging, and high safety) has set extraordinarily high standards for electrolyte materials, which are far beyond the realm of conventional nonaqueous electrolyte design based on 1 mol L-1 (M) LiPF6 and ethylene carbonate (EC). Generally, further increasing salt concentration over the conventional 1 M increases the viscosity and decreases the ionic conductivity, both of which are unfavorable for battery electrolytes in terms of reaction kinetics. However, various unusual functions have been recently discovered at high salt concentrations (over 3 M) (Fig. 1), including i) high reduction stability, ii) high oxidation stability, iii) fast electrode reactions, iv) high safety, v) wide liquidus temperature range, and vi) prevention of Al corrosion at high potentials, etc. As a result, concentrated nonaqueous and aqueous solutions are emerging as a new class of liquid electrolytes for advanced batteries. In this talk, I will introduce various unusual functions of concentrated electrolytes, which are not shared by conventional dilute electrolytes, discuss the mechanism from the viewpoint of their unique ion-solvent coordination structures, and present new electrolyte design strategies to advanced batteries.

1. Y. Yamada et al., Nat. Energy, 4, 269 (2019); 2. Y. Yamada et al., J. Am. Chem. Soc., 136, 5039 (2014); 3. J. Wang and Y. Yamada et al., Nat. Commun., 7, 12032 (2016); 4. Y. Yamada et al., Nat. Energy, 1, 16129 (2016); 5. J. Wang and Y. Yamada et al., Nat. Energy, 3, 22 (2018); 6. Q. Zheng and Y. Yamada et al., Angew. Chem. Int. Ed., 58, 14202 (2019); 7. Q. Zheng and Y. Yamada et al., Nat. Energy, 5, 291 (2020); 8. J. Wang and Y. Yamada et al., Adv. Sci., 8, 2101646 (2021); 9. S. Ko and Y. Yamada et al., Joule, 5, 998 (2021).