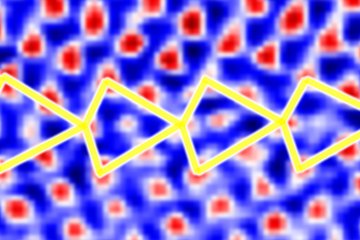
Interfacial Defects in Functional Nanostructures
Defects at interfaces strongly impact the properties and performance of functional materials. In functional nanostructures, they become particularly important due to the large surface to volume ratio.
Often, the formation of defects is energetically preferred to occur at interfaces, compared to the bulk. Such interfacial point defects are responsible, e. g., for the negative photoconductivity observed in the related project "internal interfaces in quantum dot solids". Here, we explore defects at the SiC/SiO2 interface. This interface is not only a suitable model interface, but technologically relevant for SiC-based (intermediate band) photovoltaics and power electronics.
We demonstrate that CSi carbon antisite defects at the SiC/SiO2 interface are the lowest energy defects in this system. They form preferentially at the interface, because carbon can change its hybridization from sp3 in the SiC-bulk to the lower energy sp2 state at the interface. Hence, carbon is expected thermodynamically to aggregate at the SiC/SiO2 interface.
For defects with unpaired spin states, electron paramagnetic resonance (EPR) spectroscopy is a powerful tool to identify the precise nature of the defects. Since the g-tensors measured by EPR are sensitively affected by the local chemical environment, such defects may be used as local probes for the structural properties of the interface. Based on the DFT-GIPAW approach, we compute g-tensors and hyperfine structures via spin-currents in linear response. By comparing our calculated EPR parameters to experiments, we identify common defects at the SiC/SiO2 interface and reveal why the passivation of SiC with its native oxide has remained a problem for more than 10 years.