Scope

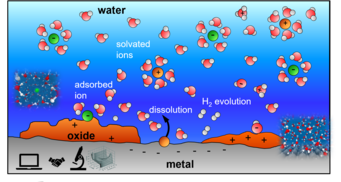
Ever since the discovery of electricity, chemical reactions occurring at the interface between a solid electrode and an aqueous solution have aroused great scientific interest, not least by the opportunity to influence and control the reactions by applying a voltage across the interface. The scientific interest continues to this day, driven by their relevance for sustainability related applications such as electrochemistry, corrosion, green hydrogen, and other. Current textbook knowledge is often based on mesoscopic concepts, i.e. effective models with empirical parameters, or focus on individual reactions decoupled from the environment, but not really sufficient to predict what happens at a particular interface under particular conditions. In the last few years both ab-initio theory and experiments have made tremendous methodological progress towards characterizing the structure and dynamics of electrified solid/liquid interfaces with atomic resolution at short timescale. We have therefore now the opportunity to achieve a truly microscopic understanding of the electrochemical interface and double layer by incorporating insights from the atomistic and electronic scale.
In order to foster this development, it is critical to bring together leading scientist from both theory and experiment to identify outstanding challenges, missing pieces, and novel directions. Our established format of tutorial-style presentations by the invited speakers, which are followed by ample possibilities for discussions allow for an in-depth insight into the various approaches and future directions. All participants will have the possibility to highlight their current research in short talks or a poster session.