Carbon partitioning during quenching and partitioning heat treatment accompanied by carbide precipitation
Carbon partitioning from martensite into austenite in the quenching and partitioning (Q&P) process has been suggested to be controlled by the constrained carbon equilibrium (CCE) criterion.

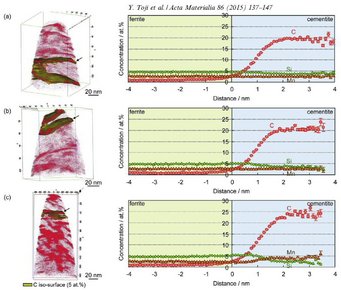
It defines an approach for predicting the carbon concentration in austenite under the condition that competing reactions such as carbide formation and bainite transformation are suppressed. Carbide precipitation in martensite is, however, often observed during the partitioning step, even in low-carbon steels as well as in high-carbon steels, even when containing a high amount of Si. Therefore, carbon partitioning from martensite into austenite is studied here, considering carbide precipitation in martensite. Carbon partitioning was investigated by means of a field-emission electron probe micro analysis (FE-EPMA) and atom probe tomography (APT), using 1.07 wt.% and 0.59 wt.% carbon steels with various martensite volume fractions. Carbon partitioning from martensite to austenite was clearly observed in all specimens, even though a considerable amount of carbide precipitated inside the martensite. The austenite carbon concentration after the partitioning step was not influenced by either the martensite volume fraction or the bulk carbon content. A modified model for predicting the austenite carbon concentration after the partitioning step was proposed to explain the experimental results by assuming carbon equilibria between austenite, ferrite and cementite under a constrained condition.