Interplay of chemistry and faceting at grain boundaries in a multicomponent engineering Al-alloy
In this project, we reveal the subtle yet important interplay between the faceting of grain boundaries and their chemical decoration with solutes in an engineering Al-Zn-Mg-Cu alloy. Previously, the interplay of chemistry and faceting was revealed for specific grain boundaries in well-defined bicrystals, which are realistically not encountered in engineering alloys.
The majority of grain boundaries in bulk metallic materials from conventional processing are of a more general type. Evidence for structural transitions in such boundaries in Al-alloys is scarce, and the interplay with chemistry is not well understood. We combined cutting-edge microscopy and microanalysis techniques to show that grain boundary facets exist in polycrystalline samples, ranging in length from a few to hundreds of nanometers. We also evidenced that the local chemistry is strongly correlated with the facet character. The self-consistent co-evolution of facet structure and chemistry leads to the formation of periodic segregation patterns of 5–10 nm, or to preferential precipitation, as rationalized by atomistic simulations. This study highlights the complexity of solute segregation to grain boundaries, defects that often underpin the mechanical properties of engineering alloys.
Figure 1(a) illustrates the Σ5 grain boundary in the as-quenched Al-Zn-Mg-Cu alloy characterized by scanning transmission electron microscopy (STEM). The grain boundary contains two sets of alternating facets along the entire length, with each type highlighted by a colored line. The energy dispersive spectroscopy in Figure 1(b) shows that the facets bear roughly 300 nm lathlike precipitates enriched with Mg and Cu. Figure 1(c) reveals that the precipitate has an orthorhombic structure, which matches the S phase (Al2CuMg).

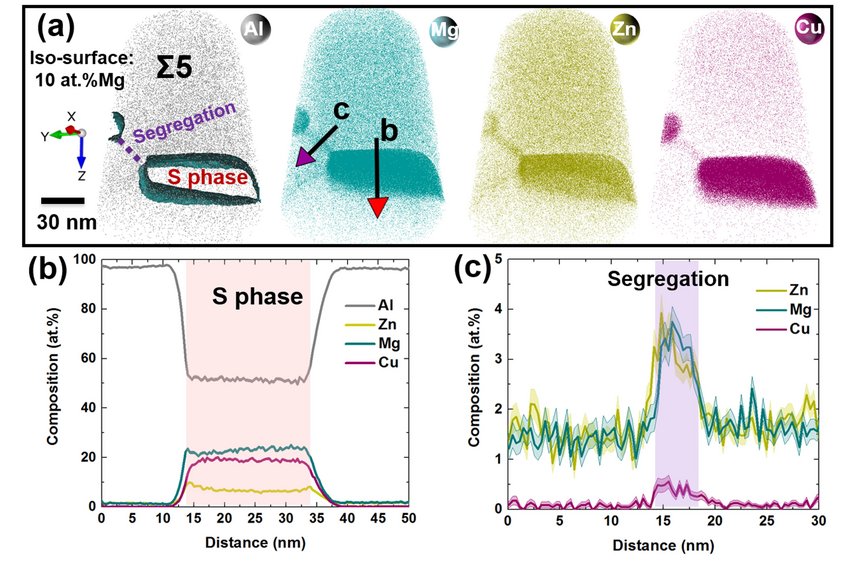
We performed complementary atom probe analyses on the same Σ5 grain boundary on another location, shown in Figure 2. Two lathlike S phases enriched in Mg and Cu are visible in Figure 2(a). The composition profile across the large precipitate in Figure 2(b) shows average compositions of 52 at.% Al, 23 at.% Mg, 18 at.% Cu and 7 at.% Zn within the precipitate. At this more detailed scale, we also observe Zn, Mg, and Cu enrichment along the {130} grain boundary facet that connects the two precipitates, and the corresponding composition profile is plotted in Figure 2(c).