Influence of carbon on stacking fault energy
High manganese steels are a major focus of research activities at the MPIE because of their high strength and ductility. These properties are crucial for the safety of modern cars. Detailed understanding of mechanisms occurring on the atomic scale during transformation processes is of main interest.
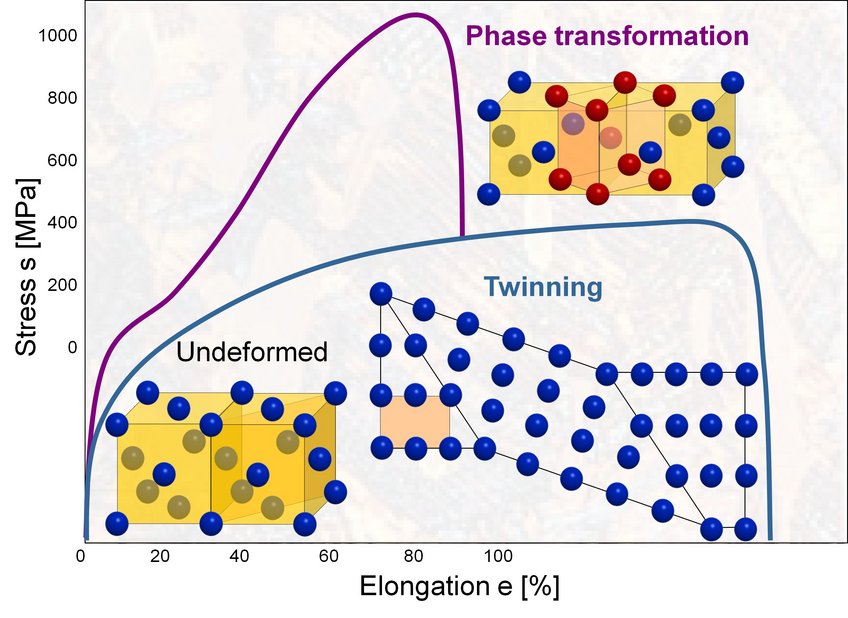
Depending on their chemical composition high manganese steels can compensate mechanical loads by sophisticated mechanisms on the atomic scale: on the one hand, twins can be formed in the crystal lattice (TWIP effect = twinning induced plasticity). On the other hand, the crystal structure can change locally from austenite to martensite (TRIP effect = transformation induced plasticity). Which of these effects occur depends on the energy required to shift the atomic layers with respect to another: the stacking fault energy (SFE).
The role of carbon in the SFE
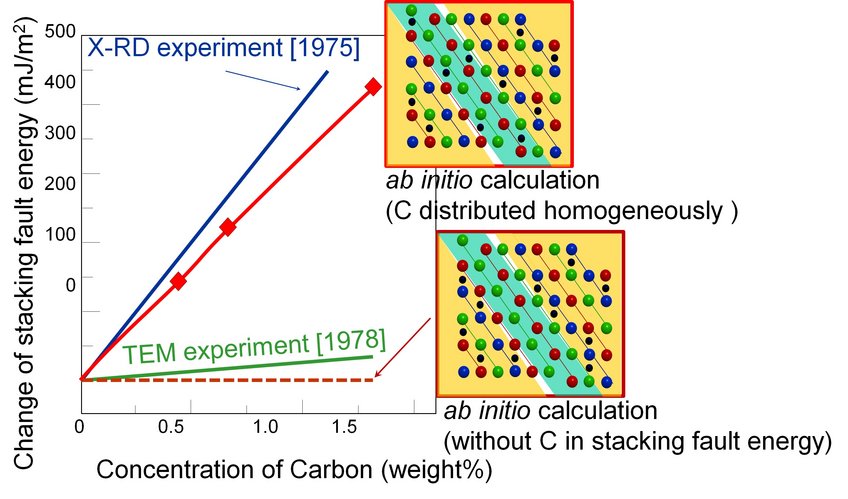
High manganese steels usually have a carbon content of up to 1 wt.%, but in the literature contradictory data are reported for the dependence of the SFE on the carbon concentration. TEM experiments show only little correlation whereas XRD indicates a strong correlation between SFE and carbon content. With novel theoretical methods developed in the department Computational Materials Design of Prof. Jörg Neugebauer, the physics of steel can be modelled accurately on the computer. With ab initio methods (ab initio = starting from the beginning) scientists can calculate energies of crystal lattices without any experimental input.

Stress-strain curves for steels showing the TRIP and TWIP mechanism.
By solving an approximation of the quantum-mechanical Schrödinger equation, which is the basis of ab initio methods, information about the physical, chemical and electronic origin of atomic interactions is obtained. “The numerical complexity is enormous”, states Dr. Tilmann Hickel, group leader in the department, “but we can manage systems of up to several hundred atoms now.” And calculations in Hickel’s group indeed show that there is a strong increase of the SFE with the carbon content. It is a definite advantage of ab initio methods that one can consider single effects separately, which is often not possible with experiments. Nevertheless, experimental validation of the theoretical results was highly desirable.
Theory and experiment go hand in hand
The ab initio calculations were per-formed under the assumption that carbon is homogeneously distributed in the crystal structure. This assumption is correct at room temperature, where carbon cannot diffuse between iron and manganese atoms.
Things change, of course, when an electron beam hits the surface during TEM measurements. This can easily transfer energy to the carbon atoms, causing a local temperature increase of up to 200-350 °C and drastically changing the diffusion rate of carbon atoms. Ab initio calculations showed that stacking fault regions caused by twinning are energetically less favourable environments for carbon, so at higher temperatures carbon diffuses a few atomic layers away and thus its influence on the SFE is diminished. This diffusion is only feasible if the energy of the carbon atoms is high enough to overcome the kinetic barriers, as is the case if the surface is bombarded with a high-energy electron beam during TEM measurements. Under normal conditions, as assumed in the calculations, the carbon atoms are immobile.

Data obtained from experiments and ab initio calculations on the dependence of the SFE on the carbon content.
In order to prove this hypothesis, TEM measurements were performed by Dr. Stefanie Sandlöbes in which the samples were cooled with liquid nitrogen. As soon as the cooling was removed, the SFE clearly decreased, which proves the hypothesis. Repeated cooling did not change the SFE value, because the carbon atoms do not return to their former positions, which remain energetically unfavourable. The experimental validation was thus successful and proves once more the efficient combination of experiment and theory.