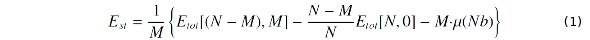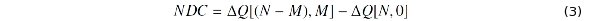Intrinsic Electronic Interactions in TiNb Alloys
Ti alloys are widely used in industry due to their superior properties, such as high strength, corrosion resistance, biological compatibility. Here, we study the substitution energy of Nb in Ti and reveal important consequences for the stability of various (meta)stable phases.
Motivation

Titanium alloys are widely used in industry due to their superior properties, such as high strength, corrosion resistance, biological compatibility. It is well known that these properties can be tuned by changing the content of the alloying elements (e.g. Nb, V, Zr, Ta, …). However, the relevant underlying physical processes are not fully understood and knowledge about intrinsic electronic interactions in Ti alloys is a prerequisite for further advances. TiNb alloys are typical members in the Ti alloy family sharing a lot of common properties/phenomena with other members (e.g. TiV, TiZr, …). Therefore, any basic law found in TiNb can be expected to be applicable to other Ti alloys. Our aim in this project is to identify the Nb-Nb and Nb-Ti interactions in Nb-doped Ti, and to understand the composition dependence of the stability of TiNb alloys. We expect that this approach in addition to our complementary work in the High accuracy ab initio prediction of phase transitions in Ti and TiNb alloys project will lead to a systematic understanding, prediction, and design of tailored Ti alloys.
Theory
To determine the energy required for/gained by alloying, we define the substitution energy (Est) per Nb atom by

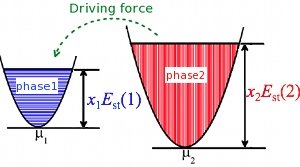
where N is the number of all atoms in the supercell, M the number of Nb atoms, Etot[(N-M),M] the energy of a Ti supercell with M Ti atoms substituted by M Nb atoms, Etot[N,0] the energy of a pure Ti supercell, and μ(Nb) the chemical potential of Nb in the BCC structure. An exothermic (endothermic) alloying process has a negative (positive) Est. The relative stability between two phases (e.g. phase 1 and 2) can be described by their cohesive-energy difference (ΔEc; see Fig. 1), which can be expressed by Est as

To identify the effect of an alloying atom on the charge density, we define also the net differential charge (NDC) by

where ΔQ is the differential charge caused by the binding between atoms.
Methodology
We use density functional theory to calculate the structural energetics and electronic properties. The computational parameters (e.g. energy cutoff and k-point grid) are systematically tested, and the energy is converged to within 1 meV. To speed up the computation without loss of accuracy, we use a special scheme in which the total energy is split into three terms

where E0 is the energy for the unrelaxed structure, ΔErel the energy change due to internal relaxation (atomic positions), and ΔEopt due to volume optimization. The calculation of the first term requires a high level of computational accuracy, while the two latter converge already at relatively low parameters. Using this scheme, the structure and energy can be quickly yet accurately calculated.
Results and Conclusions
The substitution energy (Est) sensitively depends on the structural phase of titanium (alpha, beta, omega), while it is largely independent of the Nb-Nb distance unless the Nb atoms are nearest or next nearest neighbors (see Fig. 2). This indicates that the effect of the Nb atoms is effectively screened by the electron sea, and that the Nb atoms only can interact with each other within a short range (< 3 Angstrom). This short-range interaction mechanism is directly reflected by the NDC shown in Fig. 3, where the charge disturbance by the Nb atom is quite local. The substitution is exothermic only in the β phase, while the substitution in the other two phases is energetically unfavorable. This result is consistent with experiments, because alloying only takes place at high temperatures, at which only the β phase is present.


Fig. 4 shows that the addition of Nb increases the stability of the β phase. This is consistent with the common metallurgical knowledge that Nb is a β stabilizer in Ti alloys. The critical Nb content for the α-β and ω-β stability conversion is predicted to be 25% and 43%, respectively. This prediction is consistent with the experimental observation that the ω phase disappears at a larger Nb content than the α phase. Considering the inhomogeneous distribution of the alloying elements in realistic samples, these predicted critical contents are also quantitatively consistent with experimental observations.

Other group activities
● complementary project: High accuracy ab initio prediction of phase transitions in Ti and TiNb alloys
● other projects: Adaptive Structural Materials